By Lakshmi Bangalore. Mentored and edited by Michael Newman.
My father battled Parkinson’s disease for 15 years, with each passing year feeling like an eternity. During that time, my family and I witnessed his gradual decline from a witty independent man into a person who required constant care. We watched helplessly as the disease robbed my dad of his body, mind, and spirit. With no cure in sight, he felt demoralized and often expressed suicidal thoughts.
Perhaps with more research into the biological underpinnings of my dad’s disease, there might have been a cure. However, as Joshua Sanes, professor of molecular and cellular biology at Harvard University, is quick to admit, such research is hindered by the lack of adequate models to study the human brain.
“My lab uses mice to understand the cells and circuits of the human brain, but I am here to tell you that mice aren’t enough,” he said unequivocally during a panel session at the recent annual meeting of the American Association for the Advancement of Science (AAAS).
Every year millions of people experience life-altering brain diseases that have no cure because their origin remains a mystery. Sanes and other researchers in his field are exploring new, more advanced means to define the causes of brain diseases, but not without also addressing some ethical concerns.
At the AAAS session titled “Human Neural Organoids, Transplants and Chimeras: Science, Ethics, Governance,” Sanes provided insight into the latest methods for conducting human brain research, along with fellow panelists Paola Arlotta, professor of stem cell and regenerative biology at Harvard University, and Bernard Lo, medical ethicist and professor emeritus of medicine at the University of California, San Francisco.
“Today, we can quite easily take a sample of blood or skin cells from anyone and reprogram them into stem cells, which have the potential to be converted into any cell type,” said Arlotta in her opening remarks. One method, she explained, for studying the human brain — and perhaps more importantly, how disease arises — is a brain organoid, a lab-grown “3D reductionist model” of the living organ made from stem cells.
“Now we have the opportunity to understand how we get the brain that we get,” she said. “Most importantly, we now have a human model for some of the most devastating neurological diseases affecting society — and for the first time, can investigate which cell types and biological processes are affected — all essential steps toward developing potential therapies.”
Organoid research is already yielding promising results. Arlotta reported. For example, she and her team found that nerve cells known as GABAergic neurons are produced sooner than other neurons in brain organoids derived from patients with autism. They also discovered an abnormal neural activity leading to “short circuits” in such organoids.
Sanes continued by describing another approach — neural transplants — in which researchers insert human cells into the brains of non-human animals. “The human cells integrate into circuits in the developing or adult animal brains and differentiate into more mature states than currently possible with organoids,” he explained.
“Transplantation is by no means a new method, and are even being used in clinical trials,” Sanes noted. “However, what’s new is that we’re now able to transplant human neural cells derived from people with specific diseases into animal hosts, to gain a better understanding of cellular and molecular changes that can then be used to design therapeutic improvements.”
Sanes also presented a potential third solution, neural chimeras. By injecting human stem cells into a non-human host very early in its embryonic development, researchers believe that human cells could colonize the animal’s brain and form a cross-species brain hybrid.
However, Sanes reported that viable animal-human brain hybrids haven’t yet been achieved.
“Formidable technical obstacles make it hard to predict whether such chimeras can be generated,” he noted. “If they do become a viable tool, we’ll need to figure out as a society whether the benefit accrued is worth the ethical difficulties of making an animal with a ‘human’ brain. That’s a big deal. Not something one enters into lightly.”
Following up on this point, Lo discussed the ethical issues that the new research raises, such as violating boundaries between humans and non-human animals, development of consciousness in brain organoids or development of enhanced capacities and higher levels of consciousness in chimeric or transplanted non-human animals, and concerns about the welfare and possibly the rights of animals used in this research.
“Although the level of concerns about this research is currently modest and adequately addressed by existing regulatory mechanisms, if we do not start to discuss ethical concerns now, it will be difficult to do so if the field advances rapidly and experiments raise serious concerns,” said Lo.
“The important thing is to have rigorous scientific and ethical oversight at every step of the research including across international boundaries. And that kind of protection is what can keep the science going while addressing ethics,” he concluded.
Lakshmi Bangalore holds a Ph.D. in molecular, cellular, and developmental biology from Yale University. She works in scientific communications and administration at the Center for Neuroscience and Regeneration Research at Yale Medical School. Lakshmi enjoys translating scientific reports into science stories for all. She is currently a student in the M.A. in Science Writing Program at Johns Hopkins University, and a member of the National Association of Science Writers. She can be reached at lbangal1@jhu.edu
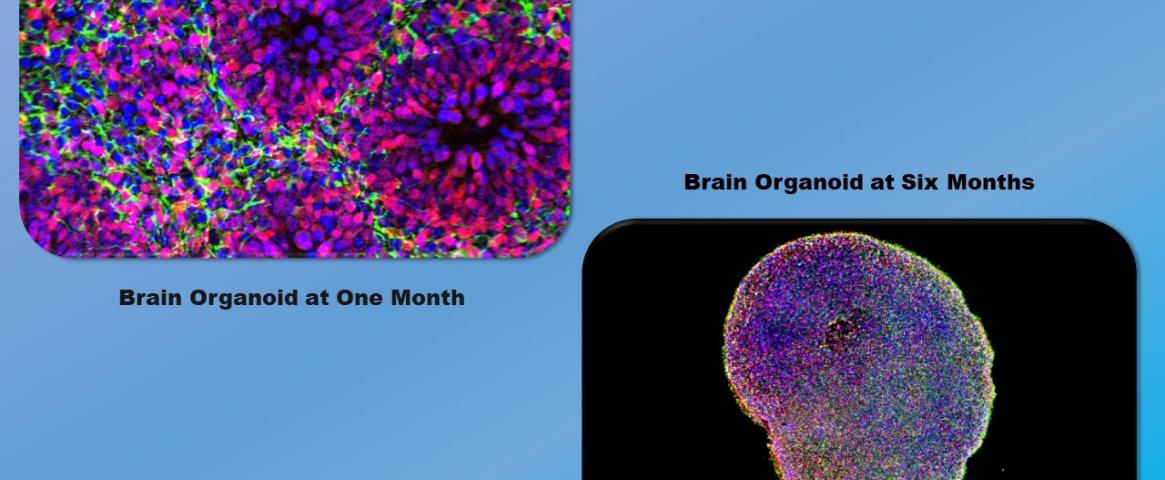
Hero image: After one month of growing in the lab (left image), a brain organoid contains clusters of cells that will develop into neurons. After six months (right image), the organoid contains multiple types of neurons (in different colors), representing the complexity of the human brain. Credit: Courtesy of Paola Arlotta Laboratory, Harvard University